
Here, we will study a general phase diagram by considering different values of one variable while keeping the other variable value constant. 
The phase diagram of a substance can be used to identify the physical and chemical properties of that substance. At this point, all three phases are interconvertible under controlled temperature and pressure values. Triple point (T) in phase diagram represents the point, where all these three curves meet and all three phases are in equilibrium due to a unique combination of temperature and pressure values. 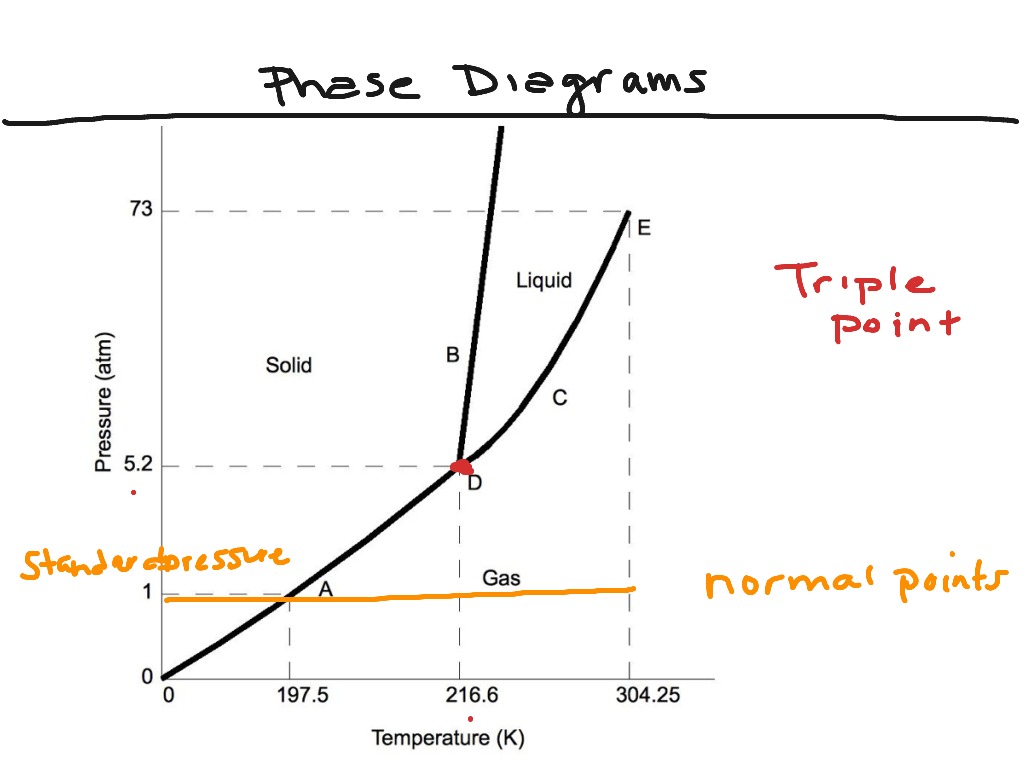
The first curve line (A, T) separated the solid and vapour phase, second curve line (T, B) separates the solid and liquid phase whereas third curve (T, C) separates the liquid and vapour phases of a substance (Fig. Triple point, as mentioned above that phase diagram has three curve lines which are also called phase boundaries. One of these points is called a critical point and the other one is called triple point.Ĭritical point (C) is explained as a point at which liquid and vapour phases of a substance are not distinguishable and the values of temperature and pressure at this point is called critical temperature and critical pressure (Fig. In a typical phase diagram, one can see two points which are very important to define in order of explaining the graph. The line of curve separates the solid and liquid phases/liquid and vapour phases/solid and vapour phases, also showing the equilibrium between two phases which means at any point on the line, two phases are interconvertible at given temperature or pressure. The first space indicates the solid phase, the second space represents the liquid while the third space shows the vapour state of a substance at a given temperature and pressure have distinct physical and chemical properties. These three spaces and three curve lines. Three spaces in phase diagram correspond to solid, liquid and vapours states of a substance. Before varying values of temperature by keeping pressure constant or vice versa we will first define certain point and features of the phase diagram. In this section, we describe how one can use the phase diagram to study the relationship between two phases and effects of varying thermodynamic conditions like pressure and temperature on properties of the substance. A phase diagram is a graphical representation of the substance phases, consists of the curved lines and the space between the two lines represent a specific phase of the matter at given pressure and temperature, whereas any point at the curve lines shows the equilibrium between two phases. A phase has a complete distinct physical and chemical properties, whereas two phases are separated by a phase boundary. Phases of a substance mean solid, liquid and gas phase of a matter. You can vary one thermodynamic condition such as temperature by keeping the other constant such as pressure. It is a very useful tool in order to understand the nature of a single component substance or mixture of substance. Phase diagram which is drawn between temperature and pressure shows the state of a substance or mixture of substances at any given time at particular temperature and pressure. Phase diagram can also be drawn between other thermodynamic conditions such as between temperature and volume or temperature and solubility etc. A phase diagram is a representation of different phases of a system consists of a substance or many substances at two different thermodynamic conditions such as temperature and pressure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
